So, in a situation like this, I think it is INACTION that generally “seals the deal” as it were. Leave a fish that’s lost its desire to swim lying on the bottom wedged under the live rock and that fish will be somewhere, dead, by morning. That’s my general prognosis. Which is why inaction is not an option. I’d rather kill the fish by trying to save it than sit back and just let it die.
Water Params in the QT Tank
Water tests came back normal…pH maybe slightly low at 8.0 but nothing alarming at all…all my reefs like to run at 8.0 most of the time. Ammonia maybe a trace, but certainly below 0.25 ppm. Nitrite and Nitrate undetectable. SG 1.010 or every so slightly higher. This, combined with the outwardly healthy and happy Lightning Maroon clownfish is an important indication that there aren’t environmental problems.
Can’t be the water…
So what is it then? Tank has had low level Formalin treatments, and I’ve followed a strict regime of Formalin dips. Tank has been at hyposaline conditions for several days, which may not outright affect a cure of any disease, but certainly should not hurt and likely has helped (I may have said it before…it could just as easily be that both fish would be dead were it NOT for hypo…we just don’t know). We’ve seen signs of Brooklynella, but certainly not the rampant infestation that takes fish down quickly. Likely I kept that at bay. There has also been Cryptocaryon. Nothing serious, certainly not to the levels where a fatality would result.
We have pretty good water parameters overall, so that’s not been a source of problems. Even got a protein skimmer going and yes, the Seaclone is pulling out foam, even at 1.010. So much for “not working” like some folks out there will tell you. Yet still, in the last 24-48 hours I have noticed elevated repiration on the Maroon Clown, and this evening, a fish on the bottom, clearly stressed. Honestly, without visual cues, the only thing that is really coming to mind is Amyloodinium, which I’ve seen before and can totally take a fish down fast. Could it be that in a week alone I may have seen all three diseases on the female Maroon?
Making Educated Guesses
Well, anything is possible. It could also be that the fish is now succumbing to starvation, secondary bacterial infections, organ failure, or even has just given up the will to live. All of those are possible.
I have to operate under the following assumptions.
1. I could STILL be facing an infection with Brooklynella.
2. I could be facing an Amyloodinium problem now.
Since neither can be comfortably ruled out at the moment, I had to think this through carefully.
Medication options for Amyloodinium vs. Brooklynella
As I’ve alluded to prior, Amyloodinium may or may not be treatable with Formalin – the literature on the subject varies, and genuinely, it is a 50/50 split. Brooklynella however, is universally cited as treatable with Formalin. Conversely, the other generally accepted treatment for parasites like these is Copper. I’m a fan of Cupramine. Seachem’s Cupramine cannot be used with Formalin – it will create a toxic substance that would probably just kill everything. Cupramine also may cause problems at hyposaline levels. No details provided, it’s a “try it at your own risk”. That said, if I was convinced I was dealing with Amyloodinium at this point, changing course to Cupramine would be a very necessary step, because Cupramine is highly effective against Amyloodinium in my experience, but only if applied early on. The other problem, Copper does nothing for Brooklynella…at least 90% of sources tend to agree on that.
If I thought (or was convinced) it was Amyloodinium?
So for the benefit of argument, what would I do if I gambled and changed to Cupramine? Well, the bottom line is any fish that’s going to be treated with Cupramine needs to be removed to another tank, because I can’t clear out the Formalin with any great level of certainty. Not worth risking a toxic cocktail. This also means possibly “rinsing” the fish as one person suggested in a comment thread. I do concur…that would be a wise precaution, and wouldn’t be that difficult to accomplish. The next thing to do would be to bring up the salinity, as the toxicity of Cupramine should decrease as that salinity goes up (a side note, we’ve also bantied about the use of an anemone as a therapeutic agent in the last 24 hours, which also would’ve required a rapid salinity change to accomplish). Some of the advisers have suggested raising salinity could in fact be done rather quickly, but their frame of reference has been largely to compare to captive bred fish. I did however, pick up good literature-based clues on the subject.
The first clue on rapid salinity rises comes from Matt Wittenrich’s suggested course of action for treating Cryptocaryon on page 161 of his Breeder’s Guide. The “jist” is that you drop SG to 1.015, and after two days, do a large water change (presumeably with full strength saltwater). But that’s not really specific. The more “detailed” clue comes from Joyce Wilkerson’s highly detailed protocol for Amyloodinium treatment on page 114 of her Clownfish Book. Again, to give only the “jist” (please see the reference for full details), drop SG to 1.010/1.012. Day 3, add copper and bring SG up to 1.018. But, “Take a day or more to bring up the specific gravity”. I’ve always worked under the assumption of no more than a 0.002 rise per day was adviseable, but this advise advoceates a rise of up to 0.008 in only 24 hours, 4X that of my assumption. Since I’m a young guy who sometimes want’s more than only the solid, highly experienced sage advice of my elders, I realized that perhaps the real kicker is something sitting in the back of my mind. Most of the bags in the Blue Zoo Aquatics shipment tested out at specific gravities around 1.019/1.020. Yet I pushed both a Cleaner Wrasse (Labroides dimidiatus) and a Harlequin Filefish (Oxymonacanthus longirostris) up to 1.025/1.026 in my reef on only a 1 hour drip. That RATE would equate to 0.120 difference over 24 hours. 60X faster than my base rate, and 15 times faster than even Wilkerson’s literature suggested to do. And I had NO PROBLEMS there.
Of course, the best example I can think of goes back to when I was 10 years old. I was breeding mollies and had my first saltwater tank. I would ROUTINELY net out my Sailfin Mollies and give them “excursions” in the saltwater tank for a day. No acclimation of any kind, just dropping them in. Now of course, I do not advocate this at all. And healthy Mollies are not the same as distressed Clownfish. But it’s just one more annecdotal experience that my preconceived notions might be ill-concieved or perhaps overly-cautious. It is clear that fish can and do withstand far more rapid rises.
So, with all that said, if I get to a point at any time where I think I need to switch to a copper-treatment for this Female Maroon, the course of action is clear. Rinse her with water from one of my broodstock reefs really quickly, and then move her to a 10 gallon tank that is set up with 5 gallons of diluted saltwater (probably from a broodstock tank). Bring the salinity up from 1.010 to 1.018, while dosing with Cupramine. That would be the way to affect a change.
However, I am not at that point yet, and I think that honestly, the time to switch would’ve been before the fish was trying to wedge itself under the live rock.
So what DID I DO?
One of the things routinely cited for treatment of both Amyloodinium AND Brooklynella is a Freshwater Dip. The FW dip is said to knock off the parasites in the case of Amyloodinium, and for anyone who’s not familiar with Amyloodinium, it can kill a fish by completely coating the gills and you may never even see it on the fish’s skin (one of the main reasons I have suspicions I’m dealing with a case of Amyloodinium now – heavy respiration, stress, and yet no think mucus – generally I’ve only seen the actual cysts on a fish when it’s commonly past the point of no return because I left the fish in the reef too long).
Formalin dips have also been good so far. Every time I did a dip, the fish always seemed “better”. Dosing the tank with Formalin this last time appeared to make things “better” as well. So I’m not about to just outright abandon this medication at this point. Especially since if I am still dealing with Brooklynella, it is THE medication to use. Amyloodnium? The jury is out, but enough people suggest it that there must be some basis for it.
The decision was to combine treatments. I admittedly have no basis for this other than the fish is now clearly on the decline and drastic steps are needed. If it’s Amyloodinium and Formalin isn’t fixing it, perhaps the FW DIP will help alleviate the stress, clear up the gills, and buy me some tmie. If Formalin IS doing the trick, why cut it out? Admittedly, I have no clue if there’s a basis for this dual purpose dip.
I had thought about doing a FW DIP with Methelyne Blue (which helps deliver Oxygen during the dip and is a often recommended practice). However, in the back of my head, I hadn’t researched it. I didn’t know how MB and Formalin would interact, and didn’t want to inadvertently cause some other problem when a fish, dipped in MB, was returned to the main tank. I have to research this more. There are potential merits.
The net result of my thinking was to use the dosage for a Formalin dip at 20 drops per gallon in a Freshwater dip to last anywhere from 5-15 minutes as tolerated (I think in the past I’ve gone even longer, but generally shorter FW dips are suggested).
Prepping the FW Dip
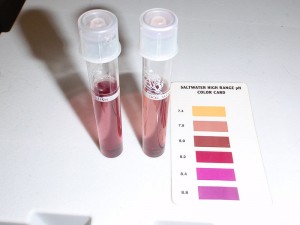
For anyone who’s never done a FW dip, it is not scary. I don’t have RO DI here unless I buy it, so I had to use good ole tap water. Matched the temperature coming out of the tap with the tank temp, and them measured out 1 gallon. Dechlorinated it with Seachem’s Prime. Then checked the pH..
The results of this first test showed that the FW from the tap (right sample) was just slightly lower than the tank’s pH. A very small dusting of Seachem’s Reef Buffer was added to the dip water, swirled and aerated for a minute. I took another test.

They are now very close, close enough in my opinion, with the FW maybe ever so slightly elevated. But you can barely tell at all. Once the pH was matched, the FW dip was “good to go.
But, as I mentioned, I opted to combine FW with Formalin. While I completed the requisite 3 dip treatment already, if I was continuing on an “every other day” Foramlin dip regime, today would’ve been the day. So 20 drops of Formalin were added to the FW dip water.
The FW + Formalin Dip
Being aware of timing on a dip is critical. Not freaking out is also critical. So, first things first, get the female Maroon out of the tank and into the dip.
Next, look at the time it is and SET AN ALARM for 10 minutes.
And now observe the fish’s behavior. No better way to convey this than video! This first video was maybe 3-4 minutes in. This is basically how the fish has been acting in the main tank since I found it this evening.
Watch, this was around the 9 minute mark…you’ll hear the alarm go off towards the end. Note how the fish makes a couple mad dashes in the bucket, and then just goes listless again. This IS the erratic behavior I mentioned earlier. The “mad dash” and then nothing.
Shortly after this vid, around 12 minutes, I felt the fish had “had enough” and returned it to the main tank. It promptly dashed around the tank and wedged itself under live rock again. But, when I left to come upstairs to document all of this, the fish was looking a tiny bit better, at least now sitting rightside up in the cave, clearly still very stressed.
EDIT – almost forgot – capped off the work with a 5 gallon water change and a full dosing of Kordon’s Fish Protector. Dosed 5 drops of Formalin as well to make up for the removed water.
But What About the Lightning Maroon?
Yeah, this IS a blog about that awesome little fish, right? Well, Edgar Diaz of AZ was the only one quick enough with the email trigger to shoot me any thoughts on my predicament this evening. He made but one suggestion in response to my many questions. To paraphrase, “Dip the female. Not the male. Female is replaceable. Male isn’t”.
It’s also come to my attention that Jake Adam’s little April 1st prank on Reef Builders STILL has a lot of people thinking that this entire fish is a myth, a photoshop job (or rather several extremely good photoshop jobs). Well, Jake has been all over me wanting dibs on the first HQ Video of the Lighting Maroon. Well Jake, it ain’t HD, it ain’t HQ, and it ain’t on Reef Builders. It’s a tease. Just enough of a tease to let everyone in the world know that this fish isn’t fabricated (unless I am now the ultimate Adobe After Effects guru, at which point you should just all bow down before me…)
Wet your appetite Jake?



Recent Comments